 
In addition, the standard can be used by other internal and external parties, such as certification bodies, to help It also covers aspects such as storage, distribution, installation and servicing, servici ng, and the provision o associated services. It is designed to be used by organizations throughout the lie cycle o a medical device, rom initial conception to production and post-production, including final decommission and disposal. ISO ����� ISO �����, Medical devices – Quality management systems – Requiremen Requirementts for regulatory regulatory purposes purposes, is an internationally agreed standard that sets out the requiremen requirements ts for a qualit qualityy management system specific to the medical devices industry.  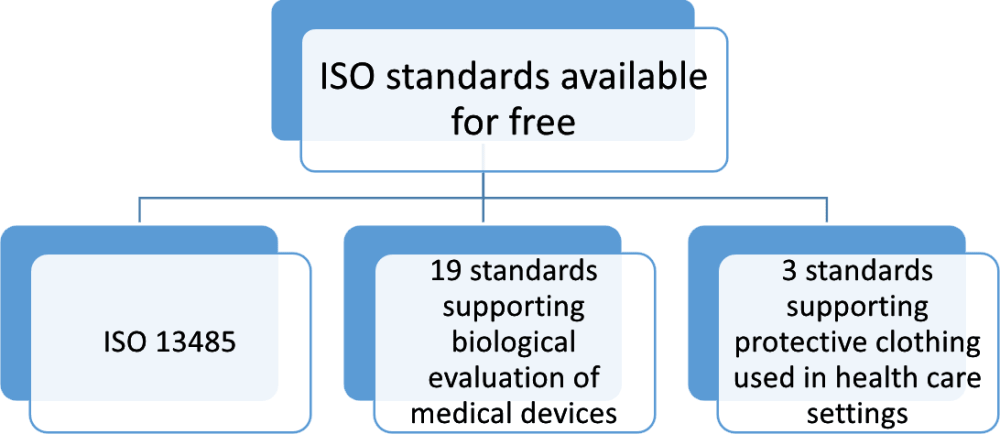
ISO ����� Quality management for medical devices
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
